Sign diaTribe's Letter to the FDA Supporting Use of CGM For Insulin Dosing
By Adam Brown
 Update (7/5/16):
Update (7/5/16):
The FDA will hold a July 21 advisory panel meeting to discuss a landmark issue in CGM: whether it should be officially approved for dosing insulin. diaTribe will be submitting a letter conveying its support for using Dexcom's G5 CGM for making treatment decisions, and the more support in the form of signatures the letter has, the more powerful it will be.
Sign our letter that will be delivered to the FDA panel members by Wednesday, July 13!
Help us reach our goal of at least 1,000 signatures on this letter; FDA has told us that there is strength in numbers and that letters with the most support carry the most weight. This letter has been reviewed and edited by over a dozen scientists and advocates within the field.
Thank you for spreading the word widely within your networks and on social media; we'd love for you to use this tweet and Facebook post ~ or any that you'd like to create ~ to make it possible for as many people to receive this and act as possible!
Sample tweet: Urge @US_FDA to approve @dexcom's G5 #CGM for insulin dosing - sign @diaTribeNews’ letter! www.diatribe.org/cgm
Sample Facebook post: Sign this letter from diaTribe + friends if you believe that the FDA should approve Dexcom’s G5 CGM as a replacement for blood glucose meters for insulin dosing! www.diatribe.org/cgm
Update (6/1/16):
The FDA just released details on the July 21 Advisory Panel hearing on whether Dexcom’s G5 CGM should be officially approved for dosing insulin (see our original article below for more information). The meeting is open to the public, and anyone interested can (i) submit a written comment to the FDA; (ii) attend the meeting; and (iii) request to give an oral presentation to the panel.
1. Submit a written comment to the FDA. All written comments must be submitted on or before July 15, 2016. Here’s how to do it:
-
Email or write your statement to Patricio Garcia at either:
-
Patricio Garcia, Center for Devices and Radiological Health, Food and Drug Administration, Bldg. 66, rm. 1116, 10903 New Hampshire Ave., Silver Spring, MD 20993
-
If emailing, please use the subject line (copy and paste the following): “FDA Advisory Panel Public Written Comment RE: CGM for Insulin Dosing”
-
Include the Meeting title and docket number:
-
Clinical Chemistry and Clinical Toxicology Devices Panel of the Medical Devices Advisory Committee; Notice of Meeting
-
Docket Number FDA-2016-N-0001
-
-
Briefly state your connection to this topic, if any (i.e., patient, healthcare provider, parent, retailer, etc.)
-
Share why you do or do not think the FDA should approved Dexcom CGM for dosing insulin (see the background below).
2. Attend the meeting in person. No registration is required:
Meeting date: July 21, 2016
Meeting time: 8 am to 6 pm
Meeting location:
Hilton Washington DC North/Gaithersburg
Salons A, B, C, and D
620 Perry Pkwy.
Gaithersburg, MD 20877
3. Request to speak during the meeting’s open public hearing (1-2 pm, July 21). The following steps must be taken on or before July 7, 2016:
-
Email, call, or write to Patricio Garcia at either:
-
Patricio Garcia, Center for Devices and Radiological Health, Food and Drug Administration, Bldg. 66, rm. 1116, 10903 New Hampshire Ave., Silver Spring, MD 20993
-
301-796-6875
-
If emailing, please use the subject line (copy and paste the following): “FDA Advisory Panel Public Oral Comment RE: CGM for Insulin Dosing”
-
Include a statement with the general nature of your presentation, your name, your address, your connection to this topic (i.e., patient, provider, etc.) and the estimated time your presentation will take (typically speakers are given no more than five minutes)
-
Oral presentations will be scheduled between 1-2 pm on July 21; if there are more speakers than the time allows, speakers will be chosen through a lottery system.
Original Article (5/9/16):
Dexcom recently announced that the FDA will hold a July 21 advisory panel meeting in Gaithersburg, Maryland to discuss a landmark issue in CGM: whether it should be officially approved for dosing insulin. The FDA’s lack of a formal approval is the key barrier to Medicare coverage – excluding hundreds of thousands of people with diabetes from using CGM.
News of this advisory panel came as a surprise, as Dexcom and the FDA have been in discussions for the last 18 months. In fact, Dexcom submitted an application last fall to update its approved labeling. Dexcom will have the opportunity to present its case to the FDA in this meeting – it has run hundreds of thousands of simulations on CGM errors, and has thousands of users in Europe where G5 is already approved for dosing insulin to help support its claim.
Why does this insulin-dosing claim matter?
-
Medicare coverage: Medicare’s obstacle to covering CGM is that it is “adjunctive” – according to the label, patients are still required to use a fingerstick meter before taking any action on a CGM reading. Many Dexcom G4 and G5 users already dose insulin off the real-time CGM readings, but the official FDA approval is what Medicare follows. An approved insulin-dosing claim does not guarantee national Medicare coverage of CGM, but it is an essential step to make it happen. (And what’s odd is that traditional blood glucose monitoring does not actually have this on its label! It still gets coverage because it is “accepted” that patients use this. We wish CGM were the same way!)
-
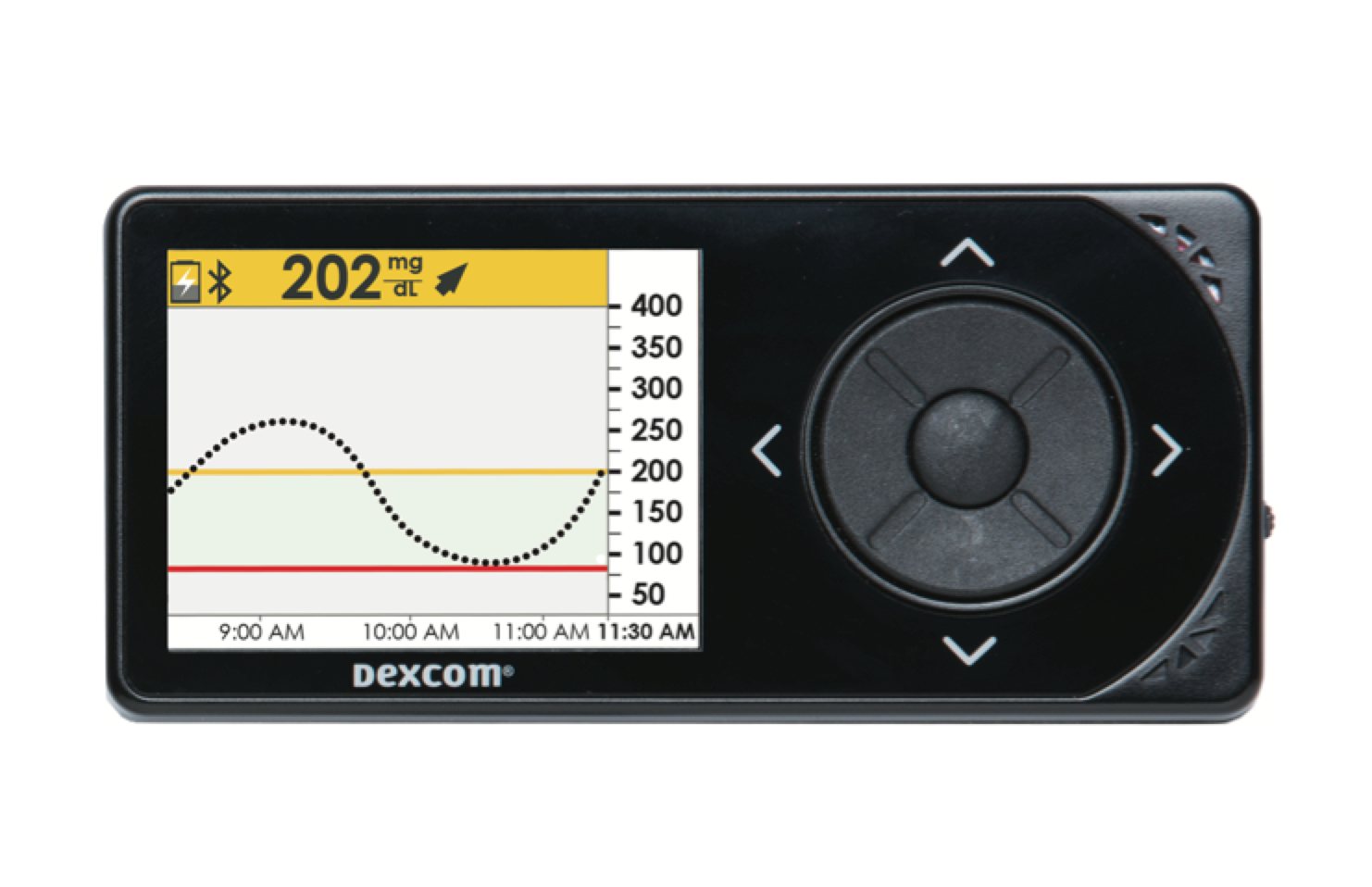
Patient safety: Dexcom’s rate of error relative to a lab standard is only 9.0%, the best in the industry, and nearly as accurate as the best glucose meters available (and probably more accurate than many low and medium quality meters). CGM also adds multiple safety features lacking with blood glucose meters: alarms, trend arrows, and minimizing how often dirty hands affect readings. The latter is very under-recognized, but can put people at risk when using just a blood glucose meter alone. While regular hand washing obviously corrects this issue, many people don’t wash their hands before taking fingersticks. In this all-too-common scenario, dosing insulin off a CGM could be safer than using a glucose meter alone.
-
Expanded Use: An approved insulin-dosing claim could expand the number of people willing to use CGM (fewer fingersticks, less hassle) and the number of doctors willing to prescribe CGM (less difficult for their patients to use). Without a dosing claim, some view CGM as just another hassle on top of using fingersticks.
-
Future products: A decision on Dexcom’s CGM insulin-dosing claim has implications for the approval of future glucose sensors, particularly Abbott’s factory calibrated FreeStyle Libre. If the FDA approves the label update for Dexcom CGM, will it also approve one for Abbott? Would Abbott’s no-fingersticks FreeStyle Libre system be required to have an insulin-dosing claim? Dexcom’s July meeting could influence how quickly FreeStyle Libre can come to US.
-
Do you believe in this indication? The diaTribe Foundation plans to collect information on this front and will be bringing members of our team and some diaTribe readers to the meeting – if you are interested, please let us know!
-AB
[Photo Credit: Dexcom]